Received: Sat 08, Aug 2020
Accepted: Thu 27, Aug 2020
Abstract
Background: Acquired uterine arteriovenous malformations (AVM) are a rare but potentially serious condition that can result in severe bleeding. Rarely, uterine AVM can occur following a complete molar pregnancy. Resolution of AVMs has been reported recently with conservative management using a combination of Gonadotropin-Releasing Hormone (GnRH) agonist, an aromatase inhibitor and tranexamic acid. This treatment has not previously been reported in a patient with AVM post molar gestation.
Case Presentation: A 23-year-old woman was diagnosed with a large uterine arteriovenous malformation following a suction curettage for a molar pregnancy. Complete resolution of the lesion on imaging was seen after six weeks of medical management.
Conclusion: A uterine arteriovenous malformation can develop in a patient post complete molar pregnancy and can be successfully treated with medical management.
1. Introduction
An arteriovenous malformation (AVM) is an abnormality in the connection between the arterial and venous networks [1]. The absence of a typical capillary network is identified when assessed by histopathology [2]. Uterine AVMs are considered rare, with true incidence unknown, but are thought to account for approximately 1-2% of uterine haemorrhage [3]. Approximately 100 cases have been published [4]. The presentation may vary from asymptomatic to severe bleeding leading to hemodynamic instability requiring hysterectomy [1]. Other possible complications can include recurrent pregnancy loss [4].
Uterine AVMs are categorized as congenital or acquired [4-6]. Congenital AVMs form due to an error in vascular differentiation and may present in multiple parts of the body [2]. The acquired subtype can occur following caesarean delivery, dilatation and curettage, or other uterine instrumentation [3, 5]. More rarely, AVM develops following cervical or endometrial cancer or gestational trophoblastic disease, infections, and even after intrauterine contraceptive device use [3, 5, 7]. Most acquired AVMs are diagnosed in women of reproductive age following pregnancy, possibly as a result of the incorporation of venous sinuses into the myometrium following necrosis of chorionic villi [1, 5, 6]. Some may also be a presentation of placental subinvolution and may spontaneously resolve [3, 6, 8].
Historically, the treatment of uterine AVMs was hysterectomy when medical management of profuse bleeding failed [4, 5]. Uterine artery embolization (UAE) continues to be used as the initial treatment in many centres despite inconsistent success rates and potentially detrimental effects on future fertility and pregnancy success [1, 5, 9]. Medical management has shown potential success in reports, especially with a combination of GnRH agonist, aromatase inhibitor and antifibrinolytic [1, 10].
Here, we describe the case of a large uterine AVM following suction curettage for a complete hydatidiform mole. The patient had full resolution of the lesion on ultrasound six weeks after conservative management with a GnRH agonist, an aromatase inhibitor and an antifibrinolytic. To the best of our knowledge, experience with this treatment in a large uterine arteriovenous malformation following molar gestation has not previously been reported.
2. Case Presentation
This case report is from The Fertility Clinic in London, Ontario, Canada. The case was exempt from Institutional Review Board review and signed consent was obtained from the patient. A 23-year-old woman, Gravida 1, Para 0 at 6 weeks gestational age presented to the emergency department with bleeding per vagina. The serum β-hCG was 8009 IU/L. Transvaginal ultrasound showed an irregular appearing intrauterine gestational sac with the mean sac diameter measuring 0.86 cm. There was no fetal pole visualized, no free fluid and no concerning adnexal or uterine masses. Three weeks later, β-hCG was 98500 IU/L and was increasing by approximately 20% every three days. Follow up ultrasound was concerning for a molar gestation, and the patient underwent a suction curettage. On pathology, the diagnosis of a complete hydatidiform mole was made.
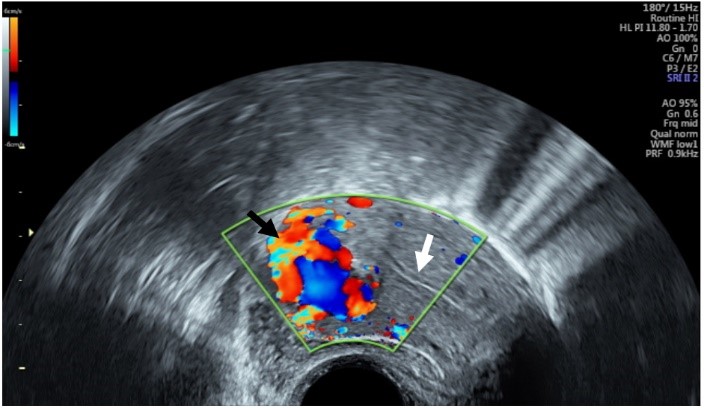
A formal ultrasound seven weeks after the suction curettage showed an anteverted uterus with a normal endometrial thickness of 0.7 cm. A heterogeneous area measuring 3.1 cm was visualized within the myometrium at the uterine fundus with prominent increased flow seen with colour Doppler ultrasound. The findings were reported as consistent with uterine AVM with no evidence of retained products of conception. The β-hCG was 1024 IU/L. Following the review of the ultrasound findings with the patient and a repeat ultrasound in the office (Figure 1), a plan was made for the attempt at conservative management with a single dose of injectable leuprolide (single 11.25 mg intramuscular injection of three months duration), tranexamic acid (1g TID for five days) and a five-day course of an aromatase inhibitor (Letrozole, 2.5mg orally, once daily) starting with the initial GnRH agonist injection, in keeping with the ABC protocol reported by Vilos et al. [1, 10], of:
i. Arrest active uterine bleeding by uterine tamponade (not needed in this case);
ii. Begin clotting of the AVM with tranexamic acid;
iii. Control the AVM with a GnRH agonist for 1-3 months;
iv. Add an aromatase inhibitor for the first 5 days of the first GnRH agonist injection to minimize the Follicle Stimulating Hormone/estrogen surge.
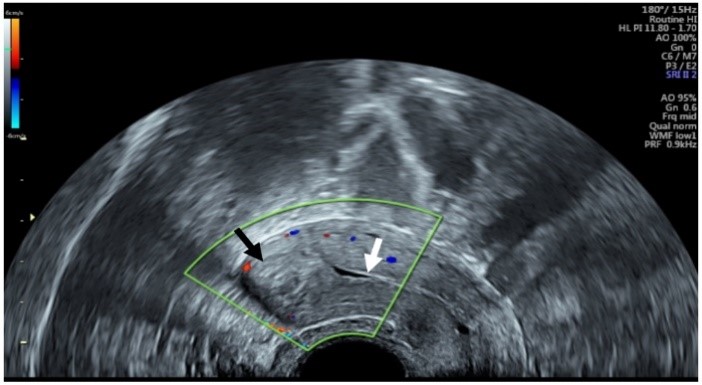
At 6-weeks post-treatment, transvaginal ultrasound showed resolution of the uterine AVM. A heterogenous appearing area was seen in the myometrium in the same location with the absence of any active blood flow on colour Doppler assessment (Figure 2). The endometrial lining was thin and distinctly separate from the area in question. The β-hCG continued to decrease steadily and was less than 1 IU/L at four months post suction curettage. The patient was having only minor spotting. The plan was to continue to follow β-hCG, and the patient was prescribed an oral contraceptive pill to prevent pregnancy for a minimum of six months of negative serum β-hCG levels. At three months post treatment, she was having normal menstrual periods and continuing on oral contraceptives.
3. Discussion
Acquired uterine AVMs generally occur in reproductive-aged women. A precursor pregnancy is thought to be important in pathogenesis [5]. They have been reported rarely following gestational trophoblastic disease, generally after chemotherapy treatment for cases of gestational trophoblastic neoplasia [7]. The disorganized trophoblastic proliferation and increased angiogenesis seen in gestational trophoblastic disease combined with the trauma of curettage used for its treatment are thought to contribute to AVM in these cases [7]. Some presentations of AVM findings on imaging are thought to reflect the subinvolution of the placental bed, a potentially transient finding [3, 6, 8]. Timmerman et al. performed an observational study of 265 patients with abnormal premenopausal bleeding [3]. Nine patients (3.4%) had features consistent with uterine AVM on ultrasound. Six of these patients proceeded to show spontaneous resolution of the changes [3].
Historically, the treatment of uterine AVM involved medical management of bleeding, blood transfusion and hysterectomy when bleeding could not be controlled [4, 5]. The majority of women with this condition are of childbearing age, and fertility potential is an important treatment consideration. Uterine artery embolization initially showed promise and continues to be used in many centers despite failure rates ranging between 17% and 60% and reports of poor future pregnancy rates and outcomes [1, 5, 9]. Some successes with a variety of conservative treatment options have been reported, including the use of danazol, methylergonovine maleate, oral contraceptives and gonadotropin-releasing hormone agonist [4]. Vilos et al., at our centre, prospectively followed 20 women with uterine AVM occurring after spontaneous or therapeutic abortion or normal pregnancy [10]. The women were treated with the same combination of a GnRH agonist, an aromatase inhibitor and tranexamic acid as our patient. All patients had complete resolution of the AVM within 1-3 months of treatment initiation, and of 16 women who attempted pregnancy, all were able to conceive spontaneously [10].
In the case reported here, the patient had complete resolution of a large acquired uterine AVM following only six weeks of treatment with injectable leuprolide. She also received an initial course of letrozole and tranexamic acid.
4. Conclusion
Uterine AVM can rarely occur following a complete molar pregnancy and treatment with a combination of GnRH agonist, an aromatase inhibitor and an antifibrinolytic was successfully used in one patient. The use of this treatment has been shown to be effective in cases of uterine AVM with reports of good future pregnancy potential.
Conflicts of Interest
None.
Consent
The patient provided signed consent to publish her case.
REFERENCES
- Vilos GA, Vilos AG, Abu Rafea B, et al. “ Resolution of uterine arterio-venous malformation followed by uneventful pregnancy after administration of gonadotropin releasing hormone agonist concomitantly with an aromatase inhibitor and tranexamic acid” Int J Womens Heal Wellness, vol. 3, no. 3, 2017. View at: Publisher Site
- Narang HK, Puri M, Patra S, et al. “Arterio-venous malformations of uterus - diagnostic and management dilemmas.” J Obstet Gynaecol, vol. 35, no. 6, pp. 632-637, 2015. View at: Publisher Site | PubMed
- Timmerman D, Van den Bosch T, Peeraer K, et al. “Vascular malformations in the uterus: ultrasonographic diagnosis and conservative management.” Eur J Obstet Gynecol Reprod Biol, vol. 92, no. 1, pp. 171-178, 2000. View at: Publisher Site | PubMed
- Peitsidis P, Manolakos E, Tsekoura V, et al. “Uterine arteriovenous malformations induced after diagnostic curettage: a systematic review.” Arch Gynecol Obstet, vol. 284, no. 5, pp. 1137-1151, 2011. View at: Publisher Site | PubMed
- Vilos AG, Vilos GA, Hollett Caines J, et al. “Uterine artery embolization for uterine arteriovenous malformation in five women desiring fertility: pregnancy outcomes.” Hum Reprod, vol. 30, no. 7, pp. 1599-1605, 2015. View at: Publisher Site | PubMed
- Cura M, Martinez N, Cura A, et al. “Arteriovenous malformations of the uterus.” Acta Radiol, vol. 50, no. 7, pp. 823-829, 2009. View at: Publisher Site | PubMed
- Touhami O, Gregoire J, Noel P, et al. “Uterine arteriovenous malformations following gestational trophoblastic neoplasia: a systematic review.” Eur J Obstet Gynecol Reprod Biol, vol. 181, pp. 54-59, 2014. View at: Publisher Site | PubMed
- Gainey HL, Nicolay KS, Lapi A “Noninvolution of the placental site: clinical and pathological studies.” Am J Obstet Gynecol, vol. 69, no. 3, pp. 558-572, 1955. View at: Publisher Site | PubMed
- Delplanque S, Le Lous M, Proisy M, et al. “Fertility, Pregnancy, and Clinical Outcomes after Uterine Arteriovenous Malformation Management.” J Minim Invasive Gynecol, vol. 26, no. 1, pp. 153-161, 2019. View at: Publisher Site | PubMed
- Vilos AG, Oraif A, Machado M, et al. “Resolution of Uterine Arteriovenous Malformation and Maintenance of Reproduction in 20 Women Treated with a GnRH Agonist Concomitantly with an Aromatase Inhibitor and Tranexamic Acid.” J Obstet Gynaecol Can, vol. 41, no. 6, pp. 772-781, 2019. View at: Publisher Site | PubMed
